Benzene Mechanism
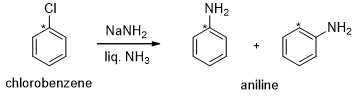
Although chlorobenzene does not contain an electron-withdrawing group, it can undergo a nucleophilic substitution reaction in the presence of a very strong base but the incoming substituent does not always end up on the carbon vacated by the leaving group. For example, when chlorobenzene is treated with amide ion in liquid ammonia, aniline is obtained as the product. Half of the product has the amino group attached to the carbon vacated by the leaving group, but the other half has the amino group attached to the carbon adjacent to the carbon vacated by the leaving group. This is confirmed by isotopic labeling method.
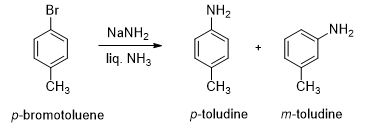
When p-bromotoluene is treated with amide ion in liquid ammonia, 50:50 mixtures of p-toludine and m-toludine is obtained.
- From the above examples one can conclude that the reaction takes place by a mechanism that forms an intermediate in which the two adjacent carbons are equivalent. The experimental observations evidence the formation of a benzyne intermediate where there is triple bond between the two adjacent carbons atoms of benzene. In the first step of the mechanism, the strong base removes a proton from the position ortho to the halogen. The resulting anion expels the halide ion, thereby forming benzyne.
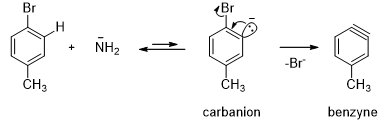
- The incoming nucleophile can attack either carbons of the “triple bond” of benzyne (Scheme 14). Protonation of the resulting anion forms the substitution product. The overall reaction is an elimination-addition reaction. Substitution at the carbon that was attached to the leaving group is called direct substitution. Substitution at the adjacent carbon is called cine (Greek: movement) substitution.
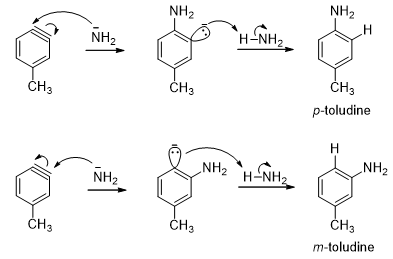
- As halide leaves with its bonding electrons from the carbanion, an empty sp2 orbital remains that overlaps with the filled orbital adjacent to it, giving additional bonding between these two carbon atoms. The two sp2 orbitals are directed 60° away from each other, so their overlap is not very effective. Triple bonds are usually linear but the triple bond in benzyne is a highly strained, so it is a very reactive intermediate. Amide ion is a strong nucleophile, attacking at either end of the benzyne triple bond. Subsequent protonation gives the product.

Reduction
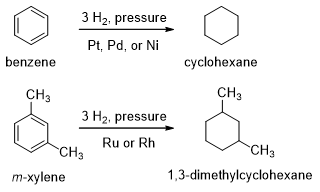
Catalytic hydrogenation of benzene to cyclohexane takes place at high temperatures and pressures. Platinum, palladium, nickel, ruthenium or rhodium is used as catalyst. The reduction cannot be stopped at an intermediate stage as these alkenes are reduced faster than benzene.
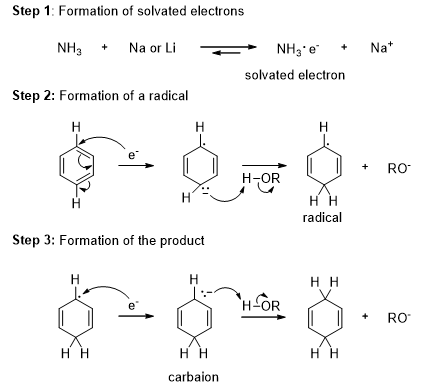
Benzene and its derivatives can be reduced to nonconjugated cyclic dienes by treating sodium or lithium liquid ammonia. This reduction is called Birch reduction.
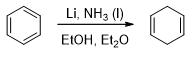
A solution of sodium or lithium in liquid ammonia contains solvated electrons that can add to benzene, forming a radical anion. The strongly basic radical anion abstracts a proton from the alcohol, giving a cyclohexadienyl radical. The radical quickly adds another solvated electron to form a cyclohexadienyl anion which is then protonated to give the reduced product.