Introduction
Atoms are connected to another homo – or hetero – atom(s) forming molecules, while, molecules of noble gases are monoatomic. This indicates that a molecule will only be formed by the combination of either homo – or hetero – atom if it tends to a low energy, filled-configuration of the outermost shell, and more stable systems than individual atomic systems.
In a di or polyatomic molecules, atoms are held together by means of an attraction force. This force is called bond. A bond could be formed by equal sharing of electron density between atoms (covalent bonding), or uneven sharing of electron density together with coulomb interaction between the atoms (ionic bonding). Whatever, a stable electronic configuration should be achieved by atom either by releasing/accepting electron(s) or sharing electron(s).
The inter-nuclear distance between two atoms is known as bond distance. The bond dissociation energy is the enthalpy change for complete splitting the units linked by a particular chemical bond in the gaseous state. It is also defined as the amount of energy released when the bond is formed between two neutral gaseous atoms.
Types of bonds
Elements can be subdivided mainly into two groups; electropositive and electronegative elements. Those elements which release or accept electron(s) to achieve an inert gas configuration are called electropositive and electronegative elements, respectively. Combination of electropositive and electronegative elements forms the following compounds.
(A) Ionic compounds = Ionic bond = Electropositive elements + Electronegative elements
(B) Covalent Compounds = Covalent bond = Electronegative elements + Electronegative elements
(C) Metallic Compounds = Metallic bond = Electropositive elements + Electropositive element
Examples
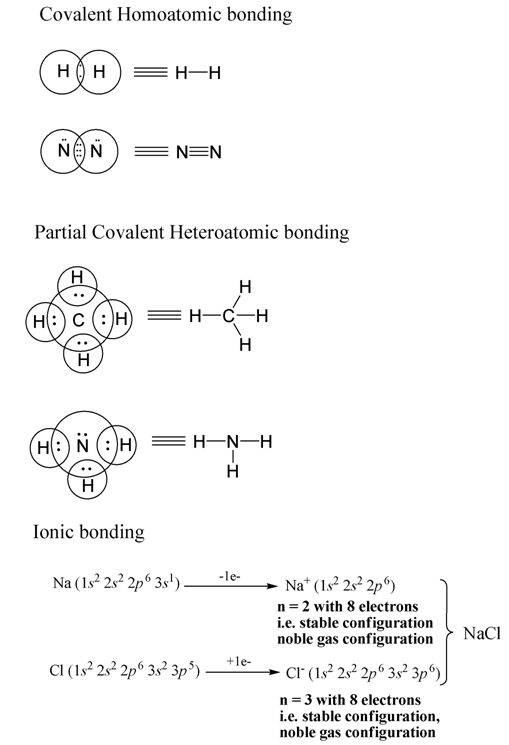
Covalent, partial covalent and ionic bonding patterns maintaining octet rule.
The ionic bond
As stated earlier, Ionic bonds are formed when electropositive elements interact with electronegative elements. The resulting compound is known as ionic compound.
Some features of ionic compounds:
(i) Ionic compounds tend to have low ionic conductivity in solid state but highly conducting in molten state. This is because in the solid state ions are held tightly by the surrounding counter ions (lattice effect) so, they are not free to move in the presence of any external electric field, whereas, in molten state it becomes easier for the movement of ions.
(ii) Ionic compounds are high melting, hard and brittle substances.
(iii) Ionic compounds are often soluble in polar solvent with high dielectric constants.
Radius ratio rules:
In ionic compounds, ions are held together by electrostatic attraction between two oppositely charged ions. The structures of ionic compounds depend on the relative size of positive and negative ions. The accommodation of the number of ions around a particular ion, i.e. coordination number, can be found out by geometric calculation.