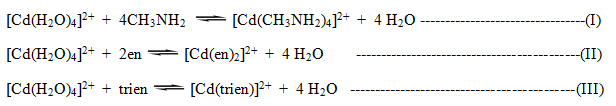The chelating effect.
Chelates
When a multidentate ligand coordinate to a metal ion from more than one donor site forming a ring with the metal, the ligand is said to be a chelating ligand and the resulting compound is said to be a chelate complex.
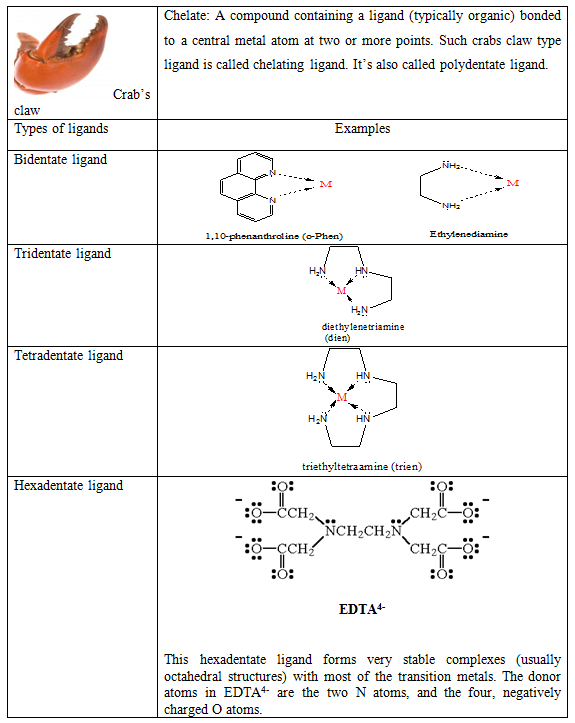

Q. Although m-Phen and o-Phen have two donor site but they are not able to behave as bidentate ligand. Why?
Hints: Due to steric reason two lone pairs at meta or para position are not able to bind with one metal atom/ion.
The chelating effect : The chelate complex es are more stable than non -chelate complexes. This effect is known as chelating effect.
Q. “Chelating effect is known as entropy effect.” Explain.
Chelating effect can be explained by considering the formation of some non-chelated & chelated complex ion of the same metal. [Cd(H2O)4]2+ form tetra coordinated complex ion with CH3NH2, en and trien.